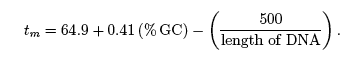Linear Functions Applications
DNA melting temperature
Imagine you have a number of different DNA samples each measuring 250 base pairs in length. Suppose that the samples are dissolved in a buffer containing 1 M NaCl and that they differ only in their GC content. If you were to calculate the melting point for the different DNA molecules, and plot it against the molecule’s GC content, you would get a line, as shown below:
In general, the linear equation used to calculate the melting point of a DNA molecule (in °C) is,
where Na+ is the molar concentration (moles/L) of sodium ions and the length of DNA is measured in base pairs (bp). In the following exercises, assume that [Na+] = 100 mM which implies the melting temperature of a DNA molecule is given by,
Use this equation to answer the following questions: Calculate the melting temperature of a DNA molecule. Determine the % GC content of a DNA molecule given its melting temperature. Compare the melting temperature of two DNA molecules. Determine the maximum melting temperature for a DNA molecule of known length. ***** |
The Biology Project > Biomath > Linear Functions > Applications > DNA melting temperature
The Biology Project
Department
of Biochemistry and Molecular Biophysics
The University of Arizona
May 2006
Contact the Development Team
http://www.biology.arizona.edu All contents copyright © 2006. All rights reserved.